MDM’s growing role in running healthcare businesses
The pharma industry has a growing number of dependencies on reliable master data

Any business doing practically anything
has a customer list. It remains a very basic, yet very challenging, part of how the pharma industry interacts with its primary customers, the healthcare providers: reliably identifying their location, credentials and affiliations (Fig. 1). For many years, the IT industry has addressed these needs (and those of other industries) with “master data management” (MDM) software. For life sciences companies, the game changed quickly to matching physician demographics with their prescribing behavior; another driver was the Prescription Drug Marketing Act (PDMA), which now requires tracking how many drug samples are delivered to a physician. In addition, corporate integrity agreements (CIAs) often specify which drugs can be sampled to which physicians.
Another driver—which the industry is still resolving—is the Physician Payments Sunshine Act, which led to the Open Payments system operated by CMS. The Sunshine Act (which, it’s worth noting, was a part of the Affordable Care Act that the current administration still wrestles with repealing) requires tying “transfers of value” (which could be a pizza delivered to a doctor’s office by a rep) to the specific physician, aggregating the spending across companies and interactions, and then making that data publicly available. From its high-drama first years of implementation (Pharmaceutical Commerce, July/Aug 2014, p. 14) to now, the annual Open Payments report has become a matter of routine, but there are significant changes going on, both in the US and abroad, over this type of transparency in pharma activity.
click to enlarge
Fig. 1. What affiliations a prescriber has affects the contractual terms of drug dispensing.
Credit: QuintilesIMS[/caption]
While these compliance requirements were evolving, so was the technology. MDM used to be looked on as basic plumbing for managing sales territories and keeping track of contact information of prescribers; now it is becoming the foundation of higher-level business intelligence and analytics, including the Big Data applications that have arisen from the large-scale communications and e-commerce like Amazon or Google. And it’s all moving to the cloud, although there is considerable debate about how best to take advantage of these technologies.
More speed
The one constant that can be discerned in both the business applications and the technology is speed of obtaining, verifying and deploying MDM data. “It’s amazing what’s happened in the past five years,” says Greg Ungemach, product specialist at MedPro Systems, one of the leading US providers of master data. “Clients were accustomed to annual or semi-annual updates of prescriber lists; now this information comes faster, from a broader set of sources, and is updated in days or even hours.” Some of this speedup is through retrieving digital data from repositories—a “machine to machine” connection, but Ungemach says that manual processes and stewardship are still a vital supplement to providing validated, reliable data.
“We track how quickly we can update a DCR [data change request] that might come in from a field rep visiting a doctor’s new location,” says Jim Cushman, VP and GM for Veeva OpenData, that company’s reference data service. “We can verify and update a record in less than five hours for 60% of DCRs, and 95% within 24 hours.” He goes on to note that in the past, the unreliability of master data often compelled users to block updates for days or weeks to allow data to be validated. “Overall data governance is better.”
Technologies are growing in number; applications are growing in number, but the vendors appear to be shrinking, especially on the data-gathering side. IMS Health (now QuintilesIMS), a longtime power in the reference data-gathering world, purchased Cegedim Relationship Management, owners of the OneKey global database in 2014; since then, it acquired Healthcare Data Solutions, another data provider, and two application-oriented firms, Privacy Analytics and Polaris Management Partners. LexisNexis, big in financial and insurance data, acquired Health Market Science in 2014. Veeva acquired AdvantageMS in 2013, and several overseas data houses. Most recently, PRA Health Sciences acquired Symphony Health Solutions—a pairing of a data provider (mostly of scrip and claims data, but also MDM reference data) and a contract research organization (CRO), just as Quintiles and IMS’ merger has linked the two.
These merged entities boast an ever-growing universe of healthcare provider (HCP) reference data: MedPro, 21 million records (primarily US); QuintilesIMS, 13 million; and Veeva, 20 million. LexisNexis claims seven billion name/address combinations (across its universe of personal and professional data). As these numbers indicate, the reference-data universe is tipping beyond HCPs and into patient databases—more about that later.
MDM + CRM
For fulfilling the traditional, basic role of an MDM—providing a sales rep with current contact information—there is a close tie-in between the MDM service and sales force automation tools, such as customer relationship management (CRM) solutions. “We got into this business because our CRM customers had problems with the data they were seeing, and blamed the Veeva CRM software,” says Cushman. Now, there can be a seamless integration between Veeva’s CRM products and its MDM reference data. Likewise, QuintilesIMS brought in both Cegedim’s CRM and its OneKey data with the acquisition; IMS Health had its own CRM platform, and has since announced new efforts in that arena. However, these and other CRM providers stress that clients have the ability to integrate other data sources of their choosing into the CRM systems.
These “other data sources” capabilities are a topic of bitter contention between Veeva and QuintilesIMS. In January, following what is said (on both sides) to be years of continually trying to resolve a data-access arrangement that would preserve proprietary QuintilesIMS data and methods, QuintilesIMS filed suit in New Jersey federal court (Case No. 2:17-cv-00177) claiming theft of trade secrets, false advertising, unfair trade practices and three other counts. Veeva then countersued, claiming antitrust violations and unfair competition.
Part of the underlying logic of QuintilesIMS’ position is the approach Veeva takes to how its reference data is updated. According to Cushman, when a Veeva client sends in a data change request (DCR), it goes through the Veeva validation process (which can include the intervention of more than 100 data stewards in the US, and a couple hundred more in offices overseas), and then becomes an updated record available to all Veeva clients. (There are contractual items involving how detailed this is; for example, the record might include an HCP’s communication channel preference, information that could be considered proprietary by the originating DCR, which would not be conveyed over to OpenData.) QuintilesIMS keeps client data contained for each client, asserting that competing pharma clients do not want to share fresh data with each other.
Third party agreements
It is possible, however, for Veeva data and QuintilesIMS data to co-exist with the same client, by means of third-party agreements (TPAs) between the various parties. “We put up a firewall such that we can push into the client’s instance of another reference data set, but that data set can’t push out to our data,” says John Busalacchi, senior principal in QuintilesIMS Enterprise Information Management center of excellence. “The client decides which reference data to use as its core.”’
QuintilesIMS’ suit maintains that these TPA have been abused; for its part, Veeva says that it provides a migration path for a current user of QuintilesIMS data to shift to Veeva’s reference data. The litigation, according to the firms, is about to enter a due diligence phase; it could go on for years.
Coming at the data validation process from another direction, LexisNexis provides what it calls a master file Data Enhancement Service; a client’s existing reference data is digitally matched against LexisNexis’ master files, and a report reconciling the two is issued. Josh Schoeller, VP, healthcare markets for LexisNexis, says that any master file degrades quickly—“physicians’ affiliations change at a rate of 30% per year, and our verification tests have shown that we can typically raise the quality of a file by 20 percentage points. We can then benchmark the quality level of the existing database, and an MDM manager can calculate an ROI in terms of improved sales team accuracy.”
MedPro (which stresses its linkage to any MDM software) highlights its linkage with Concur, a popular resource for recording and tracking travel expenses and other field-rep activities; MedPro was declared “App Center Partner of the Year” in 2017 by that firm. (Veeva and other CRM players also interact with Concur.) “Typically, a rep will record a recent doctor’s office visit in Concur, and by hitting one button, that HCP’s location and licensing information will be automatically populated with the latest MedPro data,” says Ungemach.
Although location and certifications are often considered commoditized—undifferentiated parts of MDM reference data, in fact—the topic continues to cause worry for data managers. A notable irritant is the reporting for aggregate spending under the US’s Open Payments system, and the now-established transparency standards in the EU. In the US, agg spend reporting is fairly settled; when CMS published the 2016 data on June 30, there was barely notice taken in the popular press. (ProPublica, an investigative journalism organization that had been publishing a “Dollars for Docs” report even before Open Payments, did not update its reporting this year.)
For the record, the industry reported $8.18 billion being spent on physicians and healthcare organizations and related revenue transfers; it was up by a mere 1.1% from the $8.09 billion reported in 2015. (The 2015 figure was revised based on later analysis by CMS; the original figure was $7.49 billion.) Of that, $2.8 billion was spent on “general” payments (this would include most sales-rep expenditures), but that hardly budged from the $2.68 billion spent in 2015.
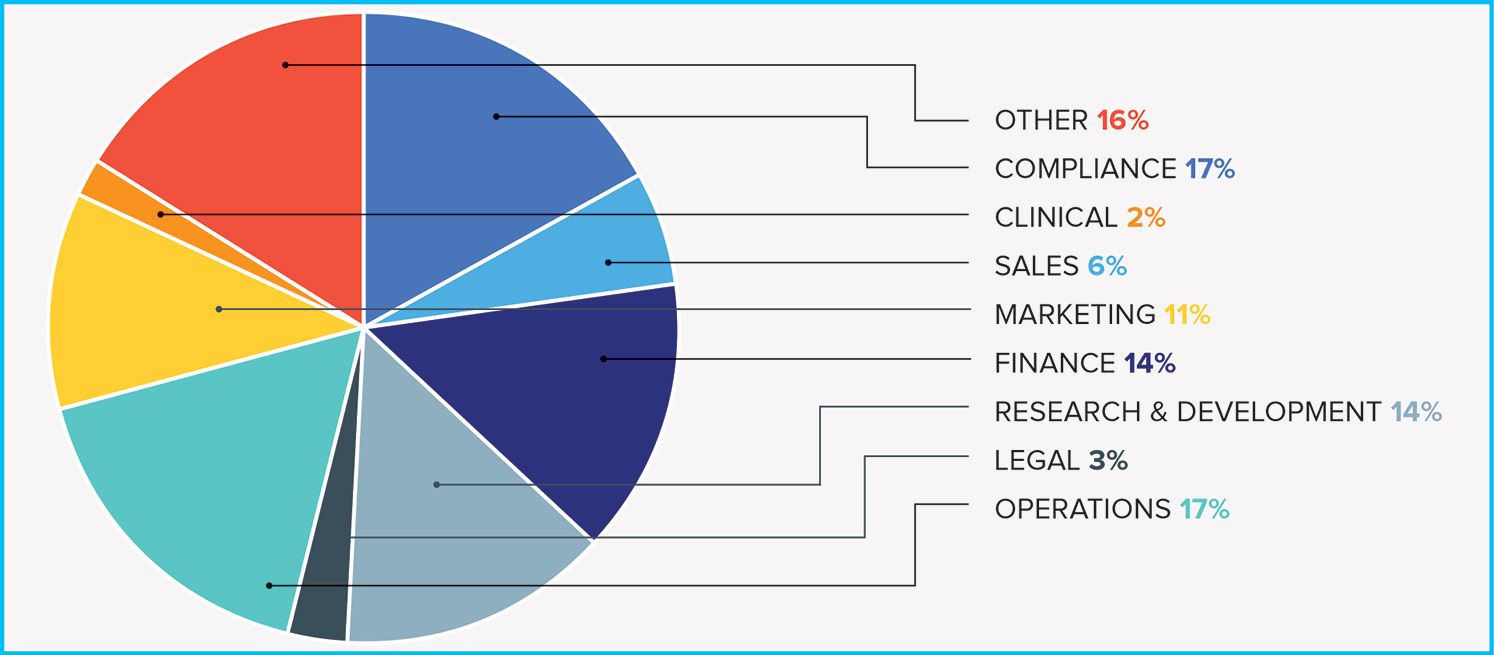
Fig. 2. Departmental affiliations of managers concerned with agg-spend reporting
A key MDM element of agg spend reporting is the National Provider Identifier (NPI) number of physicians; but here’s where such reporting gets complicated: this year, the state of Ohio established a new rule for its so-called Terminal Distributor of Dangerous Drugs (TDDD) program, which is a licensing registration for who handles most types of prescription medicines (the definition of a “dangerous drug” is one that is only available by prescription). Now, not only is the physician responsible for receiving a drug required to be identified to the state, but also the specific location where the sample was dropped off. “Doctors maintain multiple offices, so now you need to correlate who received the sample with the location of the physician,” notes MedPro’s Ungemach. “It’s essentially temporarily shut down sampling in that state until pharma companies’ databases have this information collated.”
Ohio’s rationale for changing the TDDD rules is to get a better handle on the sampling of controlled substances; however, the rule goes well beyond controlled substances. And while sampling is required to be documented nationally under the Prescription Drug Marketing Act, some states make the sample part of what gets reported as an Open Payments-type transfer of value.
Fig. 3. Technology choices for agg-spend reporting
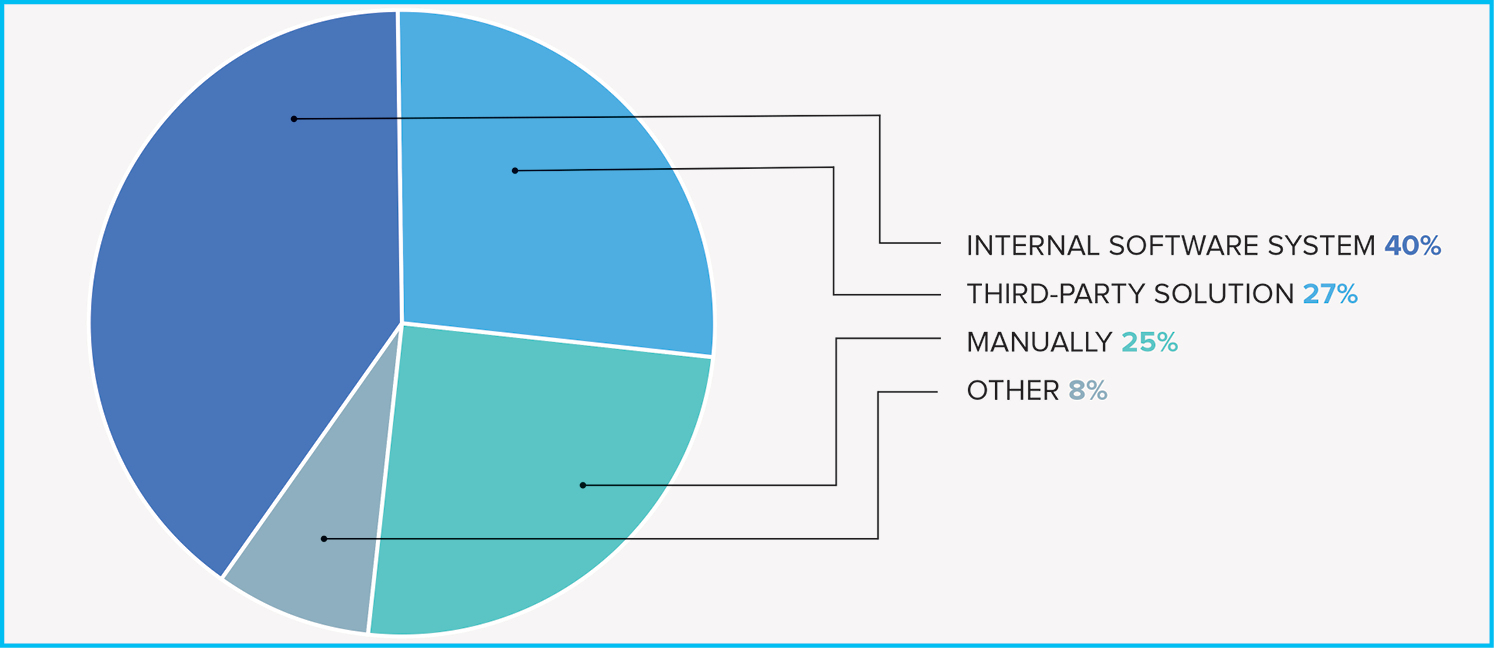
The Open Payments requirements demonstrate both how many parts of a pharma company need to track interactions with HCPS (Fig. 2) and what companies have been doing to address the need (Fig. 3). QuintilesIMS, which published a “US Trends in Aggregate Spending” survey last October, concluded that “manufacturers are not closing the book on their transparency initiatives; rather, they are just beginning the second chapter as they identify opportunities to achieve a more efficient and effective compliance operation.”
NPI numbers are just the beginning of the physician categorizations that today’s MDM systems are achieving. There are also DEA registration numbers (which used to be the default numerical ID before NPI became widely established); state medical licenses (a significant number of physicians operate in more than one state); certifications by medical specialty (which, under PDMA rules dictates what drugs can be sampled to which specialties). LexisNexis touts its sourcing of data from a consortium of 70 payers (insurers), which generates 1.6 billion medical claims annually, and whose data is used by CMS itself in tracking public insurance payments. “A good part of this work is for the benefit of healthcare providers,” says Schoeller, “but we have the opportunity to leverage this data to the pharma industry.”
The lion’s share of attention is currently focused on affiliations—which health systems prescribers belong to, and which pharmacy benefit managers and health plans have agreements with those systems. With the shift to market access teams to address prescription choices at institutional as well as individual prescriber levels, these connections become vital.
According to QuintilesIMS’ Busalacchi, affiliation data (which can also tie into related topics like formulary decisions and other factors) can best be addressed by taking an enterprise information management (EIM) perspective on MDM systems. QuintilesIMS calls this the “orchestrated customer experience,” and it could include any relevant MDM data, combined with the claims data that QuintilesIMS has been providing the industry for years, and with affiliation details as well as both structured and unstructured data (i.e., data that has not been organized into an integrated fashion). The orchestrated customer experience is branded as IMS One, but IMS One has significant technology components to it.
A key element, says Busalacchi, is a close partnership QuintilesIMS has with Reltio, a San Francisco, CA company that deploys Big Data technology to link and then visualize diverse massive data sets. According to Ajay Khanna, VP of marketing at the firm (and a former Veeva employee), one component of Reltio’s offering is a proprietary type of graphing technology (“graphing” is a term of art in organizing large data sets).
“Our graphing technology enables a user of the data to quickly pivot from, say, a view of a prescriber and that person’s affiliations, to one of the affiliations and all the other prescribers connected to it,” he says. “Additionally, a new data set or dimension can be added and the system quickly adjusts.” With conventional relational databases—or traditional MDM technology—such switching could only be carried out by reprogramming the MDM software, taking time and effort and making the maintenance of the reference data heavily burdensome.
QuintilesIMS is a value-added reseller of Reltio technology, but IMS One includes other features that QuintilesIMS is bringing together to make IMS One a “platform” for EIM, notes Busalacchi. Specific elements include Amazon Web Services such as S3, and the IMS One platform is being designed to be open to a variety of analytics services and other data-storage systems (QuintilesIMS had earlier announced relationships with Cloudera, a data-storage technology company, and with Salesforce.com, whose Force.com platform enables applications to be built quickly).
Veeva—ever ready to go at QuintilesIMS it seems—proposes a different approach. “You don’t need to bring complex analytics tools into MDM to get useful insights from it,” says Veeva’s Cushman. “You’re not going to train hundreds of field reps in data science to get a multidimensional view of their clients.” Veeva offers what is called “portlet” technology that enables data to be viewed from the MDM database, rather than generating integrations to the various other software applications. Having said that, however, Veeva’s OpenData system is accessible to those analytics systems should clients desire it. Veeva’s overall data platform is branded as Veeva Network, and Network enables easy tie-ins to other Veeva products, such as its Vault application for storing and tracking (among other things) promotional materials.
Both LexisNexis and MedPro have partnerships with Reltio, and Reltio’s Khanna says that the company aspires to be an open platform accessible to the growing array of Big Data resources now on the market.
Big data for patients
The ease with which large data sets can now be handled with cloud-based systems that offer low expense and high availability opens up another area of opportunity for the pharma industry: patient data, and dealing with patients directly.
LexisNexis’ Schoeller makes a point seldom recognized by pharma marketers: with opt-in systems, the pharma industry has a direct connection to patients; the challenge has been, what to do with it? “There hasn’t been a lot of utility to get more out of these registries than name and address standardization, but the opportunity is present to match identity data with lifestyle contexts, to get better insights into patient behaviors,” he says.
A way to address patient outcomes (without identifying specific patients) is the Systems of Care data service the company now offers, which is organized around the “social health” of patients. The service has been started initially to provide health systems with better characterization of their extended services (so that, for example, helpful lab tests can be targeted for certain therapy areas). Schoeller says that the methodology is going to be extended to patient populations during the coming year, as a potential boost for adherence programs
Veeva’s Cushman (see sidebar) notes that patient data organized in an MDM system is providing benefits to fighting the opioid crisis. And he (and others) point to the coming of wearable devices for medical monitoring, and the potential to interact with patients directly based on readouts from those devices (whether this information will flow to pharma manufacturers remains to be seen).
All of this, of course, threads the needle carefully with regard to patient privacy and HIPAA requirements. Reltio’s Khanna notes that the drive toward pay-for-performance agreements in healthcare, along with value-based contracting for pharma sales directly, is opening up channels of communication and information-sharing among payers, providers, patients and the pharma industry. It’s worth noting that Amazon Web Services, as part of its offerings to participants in healthcare, has already created resources for healthcare organizations to comply with HIPAA standards as they convey their data to cloud-based systems.
European transparency reporting
Looking for the equivalent of the US’s Open Payments system—one public repository where payments to individual HCPs is revealed and updated annually—is an eye-watering exercise in Europe. It’s not necessarily that there’s less intense public interest in the topic, but that the multinational regulatory practices across the continent allow for differing procedures, and individual privacy rights are more strongly guarded. Legislators and public interest groups who propelled the transparency initiatives had to engage in one negotiation with industry, and another with the professional organizations of HCPs, and the standards practices of those organizations.
All members of EFPIA, the European Federation of Pharmaceutical Industries and Associations (and the member companies in these associations) have agreed to provide annual reports on transfers of value to HCPs, but EFPIA does not attempt to aggregate all companies’ reports. Some countries, notably the UK, France and several other nations, have a central repository; in other cases, companies operating in the various nations report activity in each nation. EFPIA comprises 33 nations, five more than the EU.
Then there are the standards as to what’s reported. Again, there are variations, but as far as EFPIA is concerned, the key reporting on an individual basis is the funding of meetings and travel to them, speaking engagements and consulting arrangements. R&D expenditures are reported in the aggregate for each organization (other EU rules on clinical trials require identifying clinical trial investigators, but the trial funding and the investigator are apparently not combined). “Hospitality” expenditures on meals and drinks are generally not reported (considered “a disproportionate administrative burden” by EFPIA), but this varies by nation, too.
All that being said, data is beginning to come forward; EFPIA had a June 30 deadline for member companies to file reports. In the UK, the Disclosure UK program is in its second year: in June, it reported that members of the Assn. of British Pharmaceutical Industries reported a total of £454.5 million ($586.3 million) spent with HCPs and healthcare organizations—a 25% increase from 2015 (£363 million). The increase is tied to more R&D spending, said to represent 74% of total expenditures. The number of HCPs allowing individual data to be published also increased to 65% (the increased participation is not directly tied to the increased announced expenditure, which is an aggregate figure).
When it comes to individual company reports, it appears hard to beat Novartis, which has published transparency data (mostly tied to organizations, not individuals) across 38 nations. The reports are listed on one Novartis webpage, but in each country’s language and currency.
Save
Save
Save
Save
Save
Save