Drug shortages are a $360-million hit to hospital expenses, says Vizient survey
A hospital-led supply consortium is only just getting started
Drug shortages have been a persistent and mostly intractable problem for the US healthcare system for much of this century, and repeated efforts by FDA and others to address the problem have been falling short. Now, a new survey from Vizient, a GPO/health-technology provider, estimates that shortages represent a $360-million/yr hit to hospital costs, consuming 8.6 million hours of extra labor by pharmacy and administrative staff.
That cost figure is based only on the extra labor; other factors all but certainly add to it. “When you also add the cost of more expensive alternative therapies, direct purchases outside the hospital’s traditional channels, medication errors and cancelled or delayed medical procedures, we believe the actual cost of drug shortages to hospitals is significantly higher,” said Dan Kistner, SVP, pharmacy solutions for Vizient.
The survey was based on responses from 365 facilities among Vizient members, then projected to national levels. Vizient’s client base include acute and non-acute care facilities across the country, representing health systems, academic medical centers, self-governed children’s hospital, behavioral facilities, long-term care facilities, specialty hospitals and ambulatory care facilities.
Annual new drug shortages by year, according to the Univ. of Utah. credit: ASHP
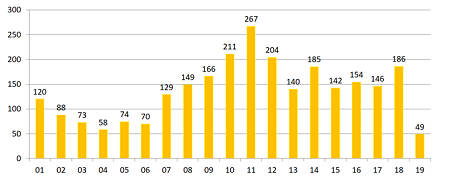
Current data (see figure) from the University of Utah Drug Information Service shows that shortages have mostly been rising since 2013; the all-time recent peak was 2011. (This service uses different criteria than FDA’s measure, but the two generally track together.) According to the survey, shortages are currently felt most critically in these categories:
- Controlled substances
- Local anesthetics
- Crash cart drugs
- Antibiotics
- Electrolytes
And respondents indicate these top-5 effects of shortages on patient care:
- Delay in medication in inpatient setting
- Delay in medication in outpatient infusion
- Delay in one or more medical procedures
- Delay in immunizations
- Cancellation of outpatient infusion
Worst of all, 38% of one or more medication errors can be attributable to a shortage situation, as providers switch out one drug for another, alter the recommended dosing of some drugs, or modify the delivery (such as from oral to injectable).
Hospitals step in
FDA has examined the problem a number of times in recent years, with studies indicating that the limited number of manufacturers for some products can cause a shortage when a manufacturing or regulatory upset occurs; generics are especially hard-hit because their pricing has been driven downward to the point where only a few manufacturers continue to operate. Some politicians have blamed FDA itself in the past, arguing that strict regulatory control makes the production of some drugs uneconomic—but this is simply another example of how drug pricing and reimbursement practices are irrational.
A year ago, a group of health systems banded together to form Civica Rx, a contracting organization to manage production of drugs in shortage directly. In May, the consortium announced its first contract, to Xellia Pharmaceuticals, a Danish developer and contract manufacturer, for two antibiotics: vancomycin and daptomycin. It has promised to have an additional 12 drugs in production by the end of this year. The drugs will be provided to consortium members (now 18 systems representing nearly 800 hospitals). Eventually Civica will be generating Abbreviated New Drug Applications (ANDAs) and perhaps even building its own manufacturing capacity.
Even so, the Civica effort will only ameliorate, not solve, the shortage problem. That the US pharmaceutical industry—operating in a market with the world’s highest drug prices—can’t address the problem meaningfully is a question looking for answers.
Multi-Indication Drug Branding in Today’s Pharmaceutical Industry
July 2nd 2025As drug development increasingly targets multiple indications, pharma companies must make strategic branding decisions—balancing regulatory requirements, market dynamics, and patient safety—to choose between single-brand or multi-brand approaches.